Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Welcome to Doctar
Join our healthcare community
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Join our healthcare community
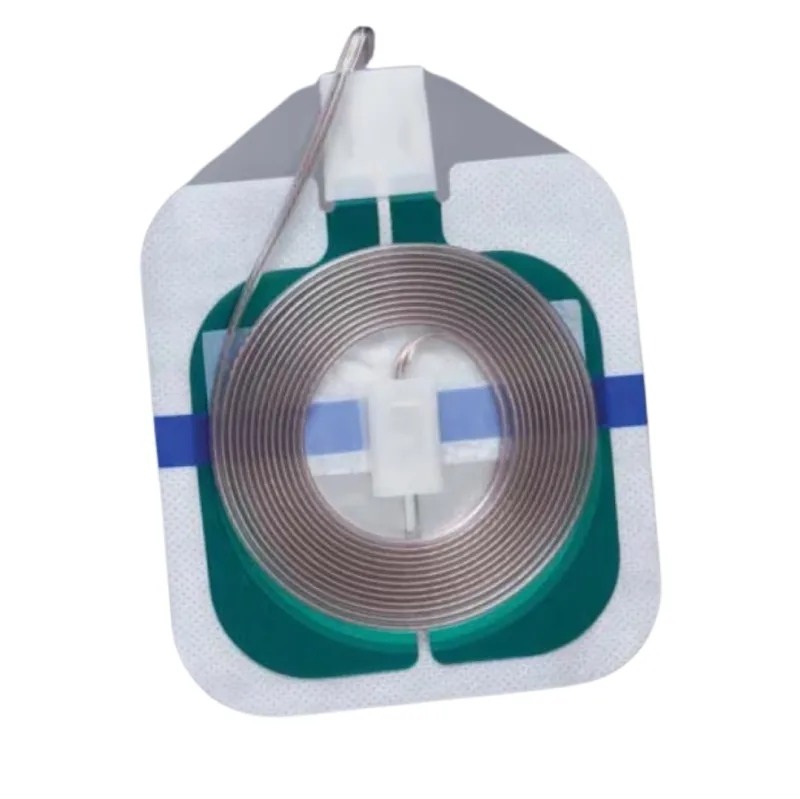
Universal electrosurgical grounding pad with split design and integrated cord for safe RF current return during surgical procedures.
Pack Size
* Prices shown are indicative. Clicking “Buy” takes you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
This is a universal electrosurgical grounding pad designed for use with electrosurgical units. It features a split design, meaning it is divided into two sections, and comes with an integrated cord for connection to the electrosurgical generator. The pad serves as a crucial component in the electrosurgical circuit, providing a safe return path for radiofrequency (RF) current, thereby preventing potential burns to the patient at unintended sites.
Its construction is engineered to facilitate efficient and secure application during surgical procedures.
The electrosurgical pad operates by establishing a conductive pathway between the patient and the electrosurgical generator. When the electrosurgical device is activated, RF current flows from the active electrode, through the patient's tissue, and then returns to the generator via this grounding pad. The pad's design is intended to distribute this return current evenly across its surface, minimizing current density and reducing the risk of thermal injury to the patient's skin.
The integrated cord ensures a reliable electrical connection throughout the procedure.
A key feature of this electrosurgical pad is its patented Safety Ring technology. This innovation is designed to mitigate the concentration of electrosurgical RF current at the edges and corners of the pad. This characteristic allows for flexible placement of the pad in any orientation relative to the surgical field, simplifying application for the healthcare provider.
Furthermore, the pad incorporates a unique transthermal backing material. This backing facilitates faster heat dissipation compared to traditional foam-backed pads, which helps to lower the risk of excessive temperature buildup beneath the pad. The transthermal backing is also engineered for tear resistance, which aids in its straightforward and clean removal after the surgical procedure is completed.
This electrosurgical pad is intended for use by trained medical professionals, including surgeons, surgical nurses, and other operating room personnel, during a wide range of surgical interventions. It is also relevant for use by anesthesiologists and other clinicians involved in patient care during surgical procedures where electrosurgery is employed. Caregivers involved in post-operative wound management may also encounter this product.
The pad is suitable for application on patients undergoing various surgical specialties, including general surgery, orthopedic surgery, cardiovascular surgery, and neurosurgery, among others.
The typical applications for this electrosurgical pad are in any surgical procedure requiring the use of a monopolar electrosurgical device. This includes procedures involving cutting, coagulation, or dissection of tissue using RF energy. The universal orientation capability makes it adaptable to diverse anatomical locations and surgical approaches.
The pad's dimensions, with an overall length of 13.2 cm and an overall width of 12 cm, are designed to provide adequate surface area for effective current dispersion in most surgical scenarios. The product is classified under code 9165.
When using this electrosurgical pad, it is important to ensure that the patient's skin is clean, dry, and free of any oils or lotions to ensure optimal adhesion and conductivity. The pad should be applied firmly to a well-vascularized area of the patient's body, away from bony prominences or areas with poor circulation, to facilitate efficient current return. The integrated cord should be securely connected to the electrosurgical generator.
Post-operative removal should be performed carefully to avoid skin trauma. Healthcare professionals should always adhere to the manufacturer's instructions for use and relevant institutional protocols for electrosurgical device safety.
Based on 2 reviews
Log in to share your experience with this product.
Log inEXCEPTIONAL SERVICE
2 May 2025
EXCEPTIONAL SERVICE
4 Mar 2025

3M
3M Tegaderm 1680 - I.V. Advanced Securement Dressing, Neonatal, 3.8 cm x 4.5 cm,