Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
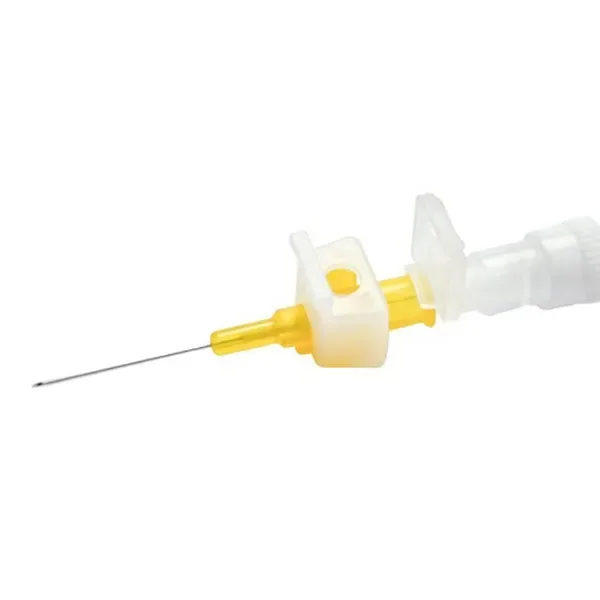
The BD Neoflon Pro IV Cannula is a medical device designed for establishing peripheral intravenous access. It is engineered to facilitate insertion into smaller and more delicate venous structures, aiming to minimize patient discomfort and potential complications. The cannula is constructed from BD Vialon biomaterial, a substance known for its ability to soften within the vein after insertion.
This characteristic is intended to improve patient tolerance during prolonged dwell times and reduce the incidence of mechanical phlebitis. The device incorporates features aimed at enhancing ease of use and safety for healthcare professionals during the cannulation procedure. Its design prioritizes secure placement and reliable venous access for the administration of fluids, medications, and for blood sampling.
The cannula is available in specific needle gauges, including 24G and 26G, to accommodate a range of patient anatomies and clinical requirements. Each unit is provided in individual packaging, with bulk options available for clinical settings.
The functionality of the BD Neoflon Pro IV Cannula is based on its design for precise venipuncture. The cannula features an anti-rotation tab, intended to provide enhanced control during insertion and offer tactile feedback to the clinician, aiding in successful cannulation. The inclusion of BD Instaflash needle technology is designed to provide immediate visual confirmation of vessel entry, potentially reducing the number of attempts required to establish access.
This visual feedback mechanism aims to streamline the cannulation process. The needle component is designed with a luer lock tip for secure connection to administration sets and includes integrated needlestick injury protection, a critical safety feature for healthcare providers. The transparent hub of the cannula allows for clear visualization of blood return, further assisting in confirming venous placement.
Key features and benefits of the BD Neoflon Pro IV Cannula include its suitability for accessing small and fragile vessels, which is a common challenge in certain patient populations, such as neonates or elderly individuals. The BD Vialon biomaterial's property of softening in vivo contributes to patient comfort and allows for extended periods of indwelling use, potentially reducing the frequency of cannula changes. The Instaflash needle technology aims to improve insertion success rates and minimize patient distress associated with multiple venipuncture attempts.
The ergonomic design and anti-rotation tab are intended to enhance procedural control and reduce the risk of dislodgement or irritation once the cannula is in place. The integrated needlestick protection mechanism is a significant safety benefit, addressing a known occupational hazard for healthcare professionals.
This IV cannula is utilized by a range of medical professionals, including nurses, physicians, and other trained healthcare providers. It is suitable for use in various healthcare environments, such as hospitals, outpatient clinics, emergency departments, and long-term care facilities. Patients requiring intravenous therapy, hydration, medication delivery, or diagnostic blood draws are the recipients of this device.
Caregivers may also interact with the device during patient care. Typical applications include the administration of intravenous fluids, antibiotics, chemotherapy, pain management medications, and nutritional support. It is also used for obtaining blood samples for laboratory analysis.
The specific needle gauges available make it particularly appropriate for pediatric and neonatal patients, as well as adults with challenging venous access.
Important usage notes for the BD Neoflon Pro IV Cannula include adherence to sterile technique throughout the cannulation procedure to prevent infection. The device is intended for single use only and must not be resterilized. It is crucial to inspect the packaging for any signs of damage or compromise before use; damaged packages should not be utilized.
The cannula should be stored in a cool, dry environment away from direct sunlight to maintain its integrity. After use, the device should be discarded immediately in an appropriate sharps container to prevent accidental needlestick injuries. The luer lock connection should be securely fastened to prevent leaks or disconnections.
Clinicians should be familiar with the specific insertion techniques and protocols relevant to their practice setting when using this device.

BD
BD 10 ML Syringe Luer Lock 21G*1 inch -100 PCS BOX

BD
BD 1ml Syringe with 27G Needle | 100 Pcs Box
BD
BD Spinal Needle

BD
BD 27G Spinal Needle

BD
BD Arterial Cannula with Flow Switch - 20G | Medical Device

BD
BD Alcohol Swab - 100 Pcs Box

BD
BD Connecta 4-Way Stopcock - IV Cannula Accessories
BD
BD 10ml Syringe Discardit - 21G*1.0 inch | 100 Pcs Box