Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
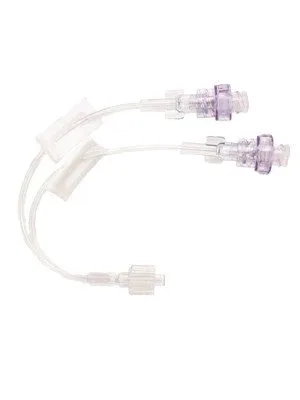
Pack
* Prices shown are indicative. Clicking "Buy" will take you to the retailer's website. Doctar may earn a small affiliate commission at no extra cost to you.
The BD Q-syte Bi-extension Needle-free Connector with Macro Bore Set and Spin Nut is a medical device designed for closed system intravenous therapy access. This connector facilitates the administration of fluids and medications while minimizing the potential for microbial contamination and accidental needle-stick injuries. The device features a split septum design that allows for easy and secure connection of luer-compatible devices, ensuring a reliable and unobstructed fluid pathway.
The macro bore tubing, measuring 15 cm (6 inches) in length, is engineered to support rapid and efficient fluid or medication delivery. The integrated spin nut mechanism provides a secure and leak-free connection to the IV administration set, enhancing patient safety and procedural integrity. This needle-free design is a key component in infection control strategies, aligning with guidelines from organizations such as the Centers for Disease Control and Prevention (CDC) and the International Nosocomial Infection Control Consortium (INICC) for the minimization of catheter-related bloodstream infections (CRBSIs).
The device has an inner diameter of 2.80mm and utilizes a male luer lock with a spin nut for secure attachment.
The operational principle of the BD Q-syte connector relies on its innovative split septum technology. When a luer-compatible device is attached, the septum creates a seal, preventing the backflow of blood and the ingress of microorganisms. Upon disconnection, the septum reseals, maintaining the closed system.
The macro bore tubing is designed to accommodate a higher flow rate, which is particularly beneficial in critical care settings where rapid fluid resuscitation or medication boluses may be required. The spin nut ensures that the connection remains secure throughout the duration of use, reducing the risk of accidental dislodgement and subsequent contamination or fluid leakage. The visible fluid channel allows for easy monitoring of fluid flow and the detection of any air bubbles or particulate matter.
Key features of this device include its needle-free design, which significantly reduces the risk of needle-stick injuries and associated infections for healthcare professionals. The split septum provides a reliable seal, and the macro bore tubing ensures efficient fluid delivery. The spin nut offers a secure and leak-free connection, contributing to the overall safety and efficacy of IV therapy.
The device is designed for straightforward integration into existing IV setups, potentially saving time for healthcare providers during procedures. The Q-syte connector is intended for single use only and should not be reprocessed or resterilized. It is supplied in sterilized packaging, with options for individual sterile packs and bulk boxes of 50 units.
This medical device is utilized by a range of healthcare professionals, including nurses, physicians, and other clinicians involved in administering intravenous therapies. It is also relevant for caregivers who may be involved in managing home-based IV treatments under medical supervision. Typical applications include the administration of fluids, medications, and blood products in various clinical settings, such as intensive care units, emergency departments, operating rooms, and general medical-surgical wards.
Its use is particularly indicated in situations where frequent access to the IV line is required for flushing, medication administration, or blood sampling, while maintaining a closed and sterile system.
Important usage notes include ensuring that the device is used only if the packaging is intact and undamaged. It is crucial to adhere to the single-use recommendation and avoid reusing or resterilizing the connector, as this can compromise its sterility and functional integrity. The device should be stored away from children.
The spin male luer lock mechanism should be tightened appropriately to ensure a secure connection without overtightening, which could damage the components. Healthcare professionals should follow established protocols for IV line management and aseptic technique when using this device to maximize patient safety and prevent complications.

BD
BD 10 ML Syringe Luer Lock 21G*1 inch -100 PCS BOX

BD
BD 1ml Syringe with 27G Needle | 100 Pcs Box
BD
BD Spinal Needle

BD
BD 27G Spinal Needle

BD
BD Arterial Cannula with Flow Switch - 20G | Medical Device

BD
BD Alcohol Swab - 100 Pcs Box

BD
BD Connecta 4-Way Stopcock - IV Cannula Accessories
BD
BD 10ml Syringe Discardit - 21G*1.0 inch | 100 Pcs Box