Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Explore ATTR amyloidosis life expectancy, symptoms, diagnosis, and treatment options. Get answers to frequently asked questions about this rare, progressive condition affecting the heart and nerves, and learn how to manage its impact on your health and prognosis.
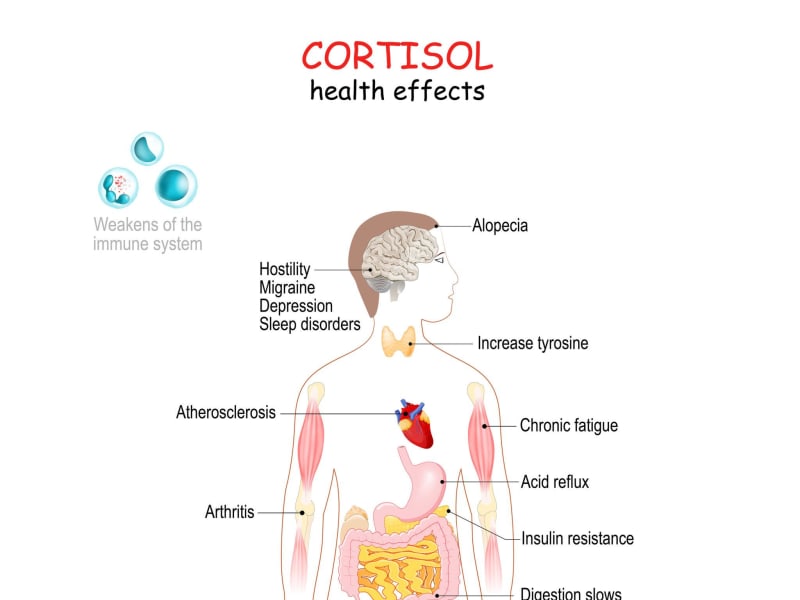
Chronic stress is now implicated in the pathology of heart disease, autoimmune conditions, digestive disorders, and cognitive decline. Understanding the biological mechanisms behind this damage is the first step in taking it seriously.
April 14, 2026

Discover how eating impacts your blood pressure, learn about postprandial hypotension, and get practical diet tips for managing blood pressure, especially for Indian readers. Includes DASH and Mediterranean diet guidance.
April 1, 2026


ATTR amyloidosis, or transthyretin amyloidosis, is a rare and serious condition caused by the misfolding of a protein called transthyretin (TTR). When TTR proteins misfold, they aggregate and deposit as amyloid fibrils in various organs and tissues throughout the body, leading to progressive damage and dysfunction. This condition can significantly impact a person's quality of life and, if left untreated, can be life-threatening. Understanding ATTR amyloidosis, its symptoms, diagnostic approaches, available treatments, and the factors influencing life expectancy is crucial for patients and their families.
Historically, ATTR amyloidosis was often underdiagnosed or misdiagnosed due to its varied symptoms mimicking more common conditions. However, advancements in diagnostic techniques and the development of targeted therapies have transformed the landscape for individuals living with this disease. This comprehensive guide aims to shed light on ATTR amyloidosis, focusing on what patients need to know about their prognosis and how to manage the condition effectively.
Transthyretin (TTR) is a protein primarily produced in the liver. Its main function is to transport thyroid hormones and vitamin A (retinol) throughout the body. In ATTR amyloidosis, this normally stable protein becomes unstable, misfolds, and clumps together to form insoluble amyloid fibrils. These fibrils accumulate in vital organs, interfering with their normal structure and function.
There are two primary forms of ATTR amyloidosis:
Both forms of ATTR amyloidosis can lead to severe organ damage, with the heart and nervous system being particularly vulnerable. The progressive nature of the disease underscores the importance of early diagnosis and intervention.
The symptoms of ATTR amyloidosis are highly variable, depending on the type of ATTR, the specific genetic mutation (in hATTR), and which organs are primarily affected. This variability often contributes to diagnostic delays. Common symptoms can be broadly categorized by the organ system involved:
Cardiac amyloidosis is a common and often life-limiting manifestation of both hATTR and wtATTR. Amyloid deposits stiffen the heart muscle, preventing it from relaxing and filling properly (restrictive cardiomyopathy).
More prominent in hATTR, amyloid deposits can damage peripheral and autonomic nerves.
Because these symptoms can overlap with many other conditions, it is critical for individuals experiencing a combination of these symptoms, especially with a family history of similar issues, to seek specialized medical evaluation.
As discussed, the fundamental cause of ATTR amyloidosis is the misfolding and aggregation of the transthyretin protein. The distinction lies in why this misfolding occurs:
This form is caused by a specific mutation in the TTR gene. Humans inherit two copies of each gene, one from each parent. In hATTR, a mutation in just one of these TTR gene copies is sufficient to cause the disease, making it an autosomal dominant genetic disorder. There are over 130 known TTR mutations, each potentially leading to a slightly different clinical presentation regarding age of onset, specific symptoms, and disease progression. For example, the Val30Met mutation is common globally and often presents with neurological symptoms, while the Val122Ile mutation is prevalent in African Americans and typically manifests as cardiac amyloidosis later in life.
If one parent has hATTR amyloidosis, there is a 50% chance that each child will inherit the mutated gene. However, not everyone who inherits the mutated gene will develop symptoms; this is known as incomplete penetrance, and the reasons for it are not fully understood.
Unlike hATTR, wild-type ATTR amyloidosis is not caused by a genetic mutation. Instead, it occurs when normal, non-mutated TTR protein spontaneously misfolds and forms amyloid fibrils. The exact trigger for this spontaneous misfolding is not fully understood, but it is strongly associated with aging. It is primarily seen in older individuals, predominantly men, typically over the age of 60, and its prevalence increases with age. This form almost exclusively affects the heart, leading to progressive heart failure, but can also cause other symptoms such as bilateral carpal tunnel syndrome.
Understanding the underlying cause (genetic vs. non-genetic) is paramount for accurate diagnosis, genetic counseling, and selecting the most appropriate treatment strategy.
Diagnosing ATTR amyloidosis can be challenging due to its diverse and non-specific symptoms, which often mimic more common conditions. The average time from symptom onset to diagnosis can be several years. A high index of suspicion, especially in individuals presenting with unexplained heart failure, neuropathy, or a combination of symptoms, is crucial.
The diagnostic process typically involves a combination of clinical evaluation, imaging, and tissue confirmation:
Historically, tissue biopsy was the gold standard for confirming amyloidosis. While the PYP scan has reduced the need for heart biopsies in ATTR cardiac amyloidosis, biopsies are still essential in certain situations:
Once ATTR amyloidosis is confirmed, genetic testing is crucial to differentiate between hATTR and wtATTR. A blood test can identify specific TTR gene mutations. This is vital for prognosis, treatment selection, and genetic counseling for family members.
A multidisciplinary approach involving cardiologists, neurologists, nephrologists, and genetic counselors is often necessary for accurate and timely diagnosis.
Treatment for ATTR amyloidosis has significantly advanced in recent years, moving beyond just symptomatic management to disease-modifying therapies that can slow or halt disease progression. The choice of treatment depends on the type of ATTR (hATTR vs. wtATTR), the primary organ involvement, and the stage of the disease.
These therapies aim to reduce the production of TTR protein or stabilize the TTR protein to prevent its misfolding and amyloid formation.
These disease-modifying therapies represent a paradigm shift in ATTR amyloidosis treatment, offering hope for slowing disease progression and improving patient outcomes.
Managing the symptoms and complications of organ damage is a crucial component of treatment, especially as the disease progresses.
The decision regarding treatment is highly individualized and requires careful consideration by a multidisciplinary team of specialists.
Understanding life expectancy in ATTR amyloidosis is complex and highly individualized. Historically, the prognosis was poor, especially for patients with significant cardiac involvement, with a median survival of only a few years after diagnosis. However, the advent of new disease-modifying therapies has dramatically improved outcomes, making past statistics less reflective of current realities.
Before effective treatments, the median survival for patients with ATTR cardiac amyloidosis was typically 2-4 years from diagnosis. For hATTR with predominantly neurological symptoms, it could range from 5-15 years. With the introduction of TTR stabilizers and gene silencers, studies have shown a significant improvement in survival. For example, tafamidis has been shown to reduce mortality in wtATTR and hATTR cardiac amyloidosis. Similarly, gene silencers have demonstrated improved neurological outcomes and reduced disease progression in hATTR polyneuropathy, potentially extending life expectancy and improving functional status.
It is important for patients and their families to have an open discussion with their medical team about their specific prognosis, considering all individual factors. While ATTR amyloidosis remains a serious, progressive condition, the future is much brighter with current therapeutic options.
Primary prevention of ATTR amyloidosis, in the sense of preventing its onset, is generally not possible because the condition is either genetic (hATTR) or age-related (wtATTR). However, there are aspects of prevention that focus on early detection and slowing disease progression:
The focus remains on early identification and aggressive management to prevent the severe complications of the disease.
Given the progressive nature of ATTR amyloidosis and the significant benefits of early intervention, knowing when to seek medical attention is crucial. You should consult a doctor if you experience any of the following symptoms, especially if they are new, unexplained, worsening, or if you have a family history of amyloidosis:
It is important to communicate all your symptoms clearly to your doctor. If ATTR amyloidosis is suspected, seek referral to a specialist center with expertise in amyloidosis, as early and accurate diagnosis is key to effective management.
A: Currently, ATTR amyloidosis is not considered curable, meaning the amyloid deposits cannot be completely removed, and the underlying cause (genetic mutation or spontaneous misfolding) cannot be reversed. However, with the advent of disease-modifying therapies, the disease progression can be significantly slowed, halted, or even improved, leading to a much better prognosis and quality of life than in the past. These treatments aim to stop further amyloid deposition and manage existing symptoms.
A: The rate of progression is highly variable. It depends on several factors, including the type of ATTR (hATTR vs. wtATTR), the specific genetic mutation (in hATTR), the age of onset, and the organs primarily affected. Some forms of hATTR can progress rapidly, while wtATTR might have a slower, more insidious onset. Early diagnosis and timely initiation of disease-modifying therapies can significantly slow down the progression and preserve organ function.
A: The main difference lies in their cause. Hereditary (hATTR) amyloidosis is caused by an inherited genetic mutation in the TTR gene, leading to the production of unstable TTR protein. Wild-type (wtATTR) amyloidosis, on the other hand, is not genetic; it results from the spontaneous misfolding of normal, non-mutated TTR protein, typically occurring in older individuals.
A: While diet cannot directly prevent or treat ATTR amyloidosis, it plays a crucial supportive role, especially in managing symptoms. For patients with cardiac involvement, a low-sodium diet can help manage fluid retention and heart failure symptoms. For those with gastrointestinal issues, dietary modifications (e.g., small, frequent meals, avoiding trigger foods) can alleviate symptoms. Consulting a dietitian specializing in cardiac or gastrointestinal conditions is often beneficial.
A: The most effective treatments are disease-modifying therapies that either stabilize the TTR protein or reduce its production. These include:
A: Yes, ATTR amyloidosis is considered a rare disease, although its true prevalence might be underestimated due to diagnostic challenges. Wild-type ATTR amyloidosis, in particular, is increasingly recognized as a cause of heart failure in older adults, suggesting it may be more common than previously thought. Increased awareness and improved diagnostic tools are leading to more diagnoses.
A: While ATTR amyloidosis primarily affects the peripheral and autonomic nervous systems and the heart, central nervous system (CNS) involvement can occur in some rare cases, particularly with certain TTR mutations and in later stages of hATTR, sometimes after liver transplantation. Symptoms might include stroke-like episodes or cognitive issues, but this is less common than peripheral neuropathy or cardiomyopathy.
ATTR amyloidosis is a complex and progressive condition that can profoundly impact multiple organ systems, with the heart and nervous system being most commonly affected. While historically associated with a poor prognosis, the landscape of ATTR amyloidosis management has been transformed by significant advancements in diagnostics and the introduction of groundbreaking disease-modifying therapies. These treatments offer real hope for slowing disease progression, preserving organ function, and dramatically improving life expectancy and quality of life for patients.
Early and accurate diagnosis is paramount. Individuals experiencing unexplained symptoms such as progressive heart failure, peripheral or autonomic neuropathy, or bilateral carpal tunnel syndrome, especially with a family history of similar conditions, should seek prompt medical evaluation. A multidisciplinary team approach at specialized amyloidosis centers ensures comprehensive care, from diagnosis to treatment and ongoing management. With continued research and accessible treatments, individuals with ATTR amyloidosis can look forward to a more optimistic future.
Explore natural ways to support blood pressure management, including foods and herbs that may act like 'natural beta-blockers'. Learn about antioxidants, L-arginine, potassium, garlic, hawthorn, red yeast rice, and omega-3s. Always consult your doctor before making changes.
April 1, 2026