Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Explore comprehensive treatments for persistent proteinuria in IgA nephropathy. Learn about symptoms, causes, diagnosis, and advanced therapies to manage kidney damage and preserve kidney function effectively.
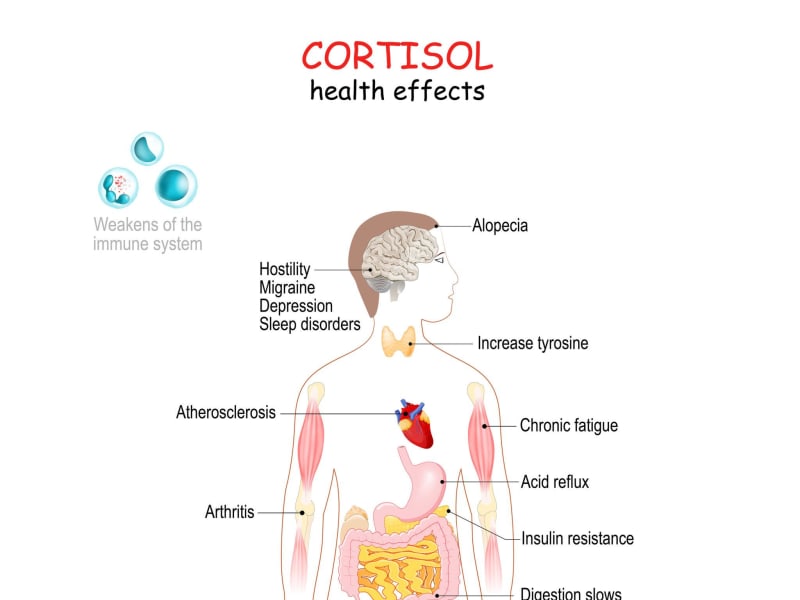
Chronic stress is now implicated in the pathology of heart disease, autoimmune conditions, digestive disorders, and cognitive decline. Understanding the biological mechanisms behind this damage is the first step in taking it seriously.
April 14, 2026

Discover how eating impacts your blood pressure, learn about postprandial hypotension, and get practical diet tips for managing blood pressure, especially for Indian readers. Includes DASH and Mediterranean diet guidance.
April 1, 2026


IgA nephropathy, also known as Berger's disease, is a common primary glomerular disease worldwide. It is characterized by the deposition of immunoglobulin A (IgA) in the glomeruli of the kidneys, which are the tiny filtering units responsible for removing waste and excess water from the blood. This deposition triggers an inflammatory response that can lead to kidney damage over time. One of the most significant indicators of kidney damage in IgA nephropathy, and a critical target for treatment, is persistent proteinuria – the presence of abnormally high levels of protein in the urine.
While some individuals with IgA nephropathy may experience a mild course with minimal symptoms, others can face progressive kidney disease, potentially leading to end-stage renal disease (ESRD) requiring dialysis or a kidney transplant. The persistence of proteinuria is a strong predictor of this progression, making its management a cornerstone of therapy. This comprehensive guide will delve into the intricacies of IgA nephropathy, its symptoms, causes, diagnostic approaches, and, most importantly, the multifaceted treatment strategies aimed at reducing persistent proteinuria and preserving kidney function.
IgA nephropathy is an autoimmune condition where the body's immune system mistakenly attacks its own kidney tissue. Specifically, it involves an abnormal form of IgA (galactose-deficient IgA1, or Gd-IgA1) that deposits in the mesangium of the glomeruli. These deposits cause inflammation and scarring, impairing the kidneys' ability to filter blood effectively. The disease can affect people of all ages, but it is most commonly diagnosed in young adults and adolescents. Its progression varies widely among individuals, from stable kidney function for decades to rapid deterioration.
Proteinuria occurs when the glomeruli, which normally prevent large protein molecules from passing into the urine, become damaged and leak protein. Persistent proteinuria, especially when exceeding 1 gram per day, is a critical biomarker indicating active kidney damage and a higher risk of kidney failure in IgA nephropathy. The amount of protein in the urine directly correlates with the severity of kidney damage and the rate of disease progression. Therefore, reducing proteinuria is a primary therapeutic goal.
IgA nephropathy is often referred to as a 'silent' disease because many people experience no noticeable symptoms, particularly in its early stages. The disease is frequently discovered incidentally during routine urine tests. However, as kidney damage progresses and proteinuria becomes more significant, symptoms may emerge.
If IgA nephropathy progresses to chronic kidney disease (CKD) or end-stage renal disease (ESRD), more severe and systemic symptoms can develop:
It is crucial to remember that the absence of visible symptoms does not mean the absence of disease. Regular medical check-ups and urine tests are vital, especially for individuals with risk factors.
The exact cause of primary IgA nephropathy is not fully understood, but it is believed to be a complex interplay of genetic predisposition and environmental factors, leading to an aberrant immune response.
Diagnosing IgA nephropathy involves a combination of tests, with a kidney biopsy being the definitive diagnostic tool.
The kidney biopsy is the gold standard for diagnosing IgA nephropathy. A small piece of kidney tissue is removed and examined under a microscope. Immunofluorescence microscopy is used to detect characteristic IgA deposits in the mesangium of the glomeruli. Electron microscopy and light microscopy further characterize the extent of kidney damage, inflammation, and scarring, which helps in staging the disease and guiding treatment decisions.
While not diagnostic for IgA nephropathy itself, imaging tests like ultrasound may be used to assess kidney size, look for structural abnormalities, or rule out other causes of kidney disease.
The primary goals of treating IgA nephropathy with persistent proteinuria are to reduce protein leakage, slow the progression of kidney damage, manage symptoms, and prevent complications. Treatment strategies are individualized based on the severity of proteinuria, degree of kidney function impairment, blood pressure, and presence of other risk factors.
These are crucial for all patients with IgA nephropathy, regardless of disease severity.
Pharmacological interventions form the core of proteinuria management.
These are the first-line therapy for proteinuria in IgA nephropathy.
These medications work by dilating the efferent arterioles of the glomeruli, which reduces pressure within the filtering units and thereby decreases protein leakage. They also have blood pressure-lowering effects and direct kidney-protective properties. They are often prescribed even if blood pressure is normal, solely for their proteinuria-reducing benefits.
For patients with persistent proteinuria (e.g., >1 g/day) despite optimal RAAS blockade and significant kidney inflammation on biopsy.
These are considered for patients with severe disease, rapidly declining kidney function, or significant inflammation unresponsive to corticosteroids.
Originally developed for diabetes, these medications have shown remarkable kidney-protective effects, including reducing proteinuria and slowing CKD progression, even in non-diabetic patients with IgA nephropathy.
A newer class of medication specifically approved for IgA nephropathy with rapid progression risk.
Some studies suggest a potential benefit in reducing proteinuria and slowing progression in certain populations, particularly those with milder disease or those unresponsive to other therapies. Evidence is mixed, and it's not a first-line therapy.
Research is ongoing for new treatments targeting specific pathways in IgA nephropathy, including:
Beyond specific IgA nephropathy treatments, managing complications is vital for overall health and kidney preservation.
For patients whose IgA nephropathy progresses to end-stage renal disease (ESRD), renal replacement therapy becomes necessary.
As the exact cause of primary IgA nephropathy is not fully understood, there is no definitive way to prevent its onset. However, there are strategies to prevent its progression and minimize kidney damage once diagnosed:
It is important to consult a healthcare professional if you experience any symptoms that might suggest kidney problems or IgA nephropathy. Early detection and intervention are crucial for better outcomes.
Regular follow-up with a nephrologist (kidney specialist) is essential for anyone diagnosed with IgA nephropathy to monitor kidney function, proteinuria, and adjust treatment as needed.
A: Persistent proteinuria means that there are consistently elevated levels of protein in your urine over a period of time, usually greater than 150 mg per day. In IgA nephropathy, it often refers to protein excretion exceeding 0.5-1 gram per day, indicating ongoing kidney damage and a higher risk of disease progression.
A: Currently, there is no definitive cure for primary IgA nephropathy. However, treatment aims to manage the disease, reduce inflammation, control proteinuria, and slow or halt the progression of kidney damage, allowing many people to live full lives with stable kidney function. Research into new therapies is ongoing.
A: IgA nephropathy is considered the most common primary glomerular disease worldwide, particularly prevalent in Asia and among Caucasians. Its prevalence varies geographically, but it accounts for a significant portion of kidney biopsies performed for glomerulonephritis.
A: While a healthy diet (low sodium, moderate protein, balanced) is a crucial component of managing IgA nephropathy and supporting kidney health, it is generally not sufficient as a standalone treatment for persistent proteinuria or progressive disease. It must be combined with prescribed medications and other lifestyle modifications under medical supervision.
A: The long-term outlook for IgA nephropathy is highly variable. About 20-30% of individuals may progress to end-stage renal disease (ESRD) within 10-20 years. However, many others will experience a stable course with preserved kidney function, especially with early diagnosis and aggressive management of proteinuria and blood pressure. Regular monitoring and adherence to treatment are key factors in determining the prognosis.
A: Yes, IgA nephropathy can recur in a transplanted kidney. The recurrence rate varies but can be significant. However, recurrent IgA nephropathy in the transplant is often milder than the original disease, and it can sometimes be managed with immunosuppressive therapy.
Persistent proteinuria in the context of IgA nephropathy is a serious indicator of ongoing kidney damage, demanding proactive and comprehensive management. While IgA nephropathy presents a variable course, from benign to aggressive, modern therapeutic approaches offer significant hope for slowing its progression and preserving kidney function. The cornerstone of treatment involves strict blood pressure control, aggressive reduction of proteinuria through RAAS blockers, and increasingly, the use of SGLT2 inhibitors and targeted immunomodulatory agents like sparsentan or targeted-release budesonide.
Lifestyle modifications, including dietary adjustments and regular exercise, play an indispensable role in supporting kidney health and augmenting pharmacological interventions. Early diagnosis, vigilant monitoring through regular urine and blood tests, and close collaboration with a nephrologist are paramount for optimizing outcomes. As research continues to unravel the complex pathogenesis of IgA nephropathy, the landscape of treatment is continuously evolving, bringing new and more effective strategies to combat this challenging kidney disease. Patients are encouraged to be active participants in their care, understanding their condition and adhering to personalized treatment plans to safeguard their kidney health for the long term.
Disclaimer: This article provides general information and does not constitute medical advice. Always consult with a qualified healthcare professional for diagnosis and treatment of any medical condition. Information from sources like Healthline (e.g., Healthline's article on persistent proteinuria treatment in IgA nephropathy) can be a useful starting point for understanding your condition, but personalized medical guidance is essential.
Explore natural ways to support blood pressure management, including foods and herbs that may act like 'natural beta-blockers'. Learn about antioxidants, L-arginine, potassium, garlic, hawthorn, red yeast rice, and omega-3s. Always consult your doctor before making changes.
April 1, 2026