Free Helpline
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
We are here to assist you.
Health Advisor
+91-8877772277Available 7 days a week
10:00 AM – 6:00 PM to support you with urgent concerns and guide you toward the right care.
Discover how automated paired blood cultures revolutionize fungemia diagnosis. This advanced test offers faster, more accurate results for critical fungal infections.

Deep within the body, a serious battle can unfold without obvious warning signs. When a fungal pathogen enters the bloodstream, a condition known as fungemia, it represents a significant medical emergency. For those affected, especially individuals with weakened immune systems, this invasion can rapidly lead to severe complications like sepsis and organ failure. The challenge for physicians has always been time — a race against an invisible clock to identify the specific microbial culprit and initiate the right therapy. How serious is this really? Mortality rates associated with certain types of fungemia, particularly those caused by Candida species, can be alarmingly high, making rapid diagnosis a truly life-saving intervention.
Traditionally, pinpointing these organisms from blood samples was a slow and meticulous process. It involved manual laboratory methods that required days, sometimes even weeks, to yield a definitive result. These techniques were not only labor-intensive but also prone to missing less common or slow-growing species.
This diagnostic delay created a dangerous gap where a critical illness could progress unchecked. Waiting for a confirmed identification often forced specialists to start broad-spectrum antifungal medications, an approach that isn't always targeted and can contribute to resistance. Every hour without a precise diagnosis matters immensely, directly impacting a person's chances of recovery.
Modern laboratories have thankfully evolved. Systems that automatically detect pathogens in blood now stand as the gold standard, fundamentally changing how we identify systemic microbial presence. These sophisticated instruments continuously monitor samples around the clock, incubating them in optimal conditions.
They work by detecting subtle changes in gas concentration (like CO2) produced by growing microorganisms, including yeasts and molds. Here's the thing — the moment such a system detects growth, it immediately flags the sample, alerting laboratory staff hours or even days earlier than manual methods ever could. This technological leap has dramatically shortened the window from sample collection to preliminary results, enabling a much faster clinical response.
The numbers don't lie.
To specifically enhance the detection of these organisms, specialists have refined the process further with a vital technique: the paired test. Instead of relying on a single sample, this approach involves collecting two sets of specimens from the person at the same time. One set is inoculated into standard aerobic and anaerobic bottles designed for general bacterial growth.
The other, key set is placed into a specialized mycotic bottle. This dual-pronged strategy is a game-changer for diagnosing fungemia.
The secret lies in the unique formulation of the media. The specialized bottle contains a nutrient-rich broth specifically designed to support the growth of various yeasts and molds.
Crucially, this medium is often supplemented with antibacterial agents that suppress any competing bacteria in the specimen. By creating an exclusive environment for these microbes to thrive without bacterial interference, it significantly boosts the chances of isolating the true pathogenic cause of the illness.
Using this paired method delivers several crucial advantages. You'll notice that these organisms often grow faster and more reliably in their dedicated bottle, leading to a quicker positive signal from the detection system. This approach also improves the recovery rate of fastidious microbes — picky organisms that might not grow well in standard media. What most people miss: this technique also assists physicians in distinguishing a genuine illness from a simple skin contaminant. If a microbe like Candida grows only in the specialized bottle but not the standard ones, it strengthens the evidence for a true bloodstream presence. So what does that mean for you? It means your care team gets a clearer, more confident diagnosis faster, allowing them to switch from general to highly targeted therapy with precision.
While any suspected systemic illness warrants investigation, the paired test is particularly crucial for certain high-risk groups. Its use is prioritized for people who are most vulnerable to invasive disease from these organisms.
Worth knowing: this includes critically ill individuals in the Intensive Care Unit (ICU), cancer patients undergoing chemotherapy, and organ transplant recipients on immunosuppressive drugs. Others who benefit are those with long-term indwelling central venous catheters or people who have been on broad-spectrum antibiotics for extended periods, as these factors can disrupt the body's natural microbiome and create an opening for microbial invaders.
That's the part worth remembering.
For those undergoing the examination, the process is straightforward and focuses on precision from the very start.
A healthcare professional will draw specimens, typically from two different sites on your body (e.g., one from each arm). This is done using a strict aseptic technique to prevent skin bacteria from contaminating the sample and causing a false positive. Each draw is then divided between the standard and the specialized mycotic bottles.
Once in the lab, the bottles are immediately loaded into the detection system. The machine takes over from there — gently agitating and warming the bottles while its sensitive detectors continuously scan for any sign of microbial life. This process runs 24/7 without interruption.
As soon as a bottle is flagged as positive, a small amount of the specimen is examined under a microscope for an initial look at the organism. A portion is also grown on a solid medium to produce a pure colony. From there, advanced technologies like MALDI-TOF mass spectrometry or molecular testing can identify the exact species of microbe within hours.
Following identification, susceptibility testing (AFST) is performed. This vital final step tells your physician precisely which medications will be most impactful against your specific illness, ensuring your therapy is as effective as possible.
Here's where it gets interesting.
Ultimately, this paired examination is more than just a diagnostic tool; it is a cornerstone of modern disease management. This reliable and rapid approach supports specialists in making swift, evidence-based decisions, directly contributing to enhanced outcomes and offering a fighting chance to people facing these serious illnesses.
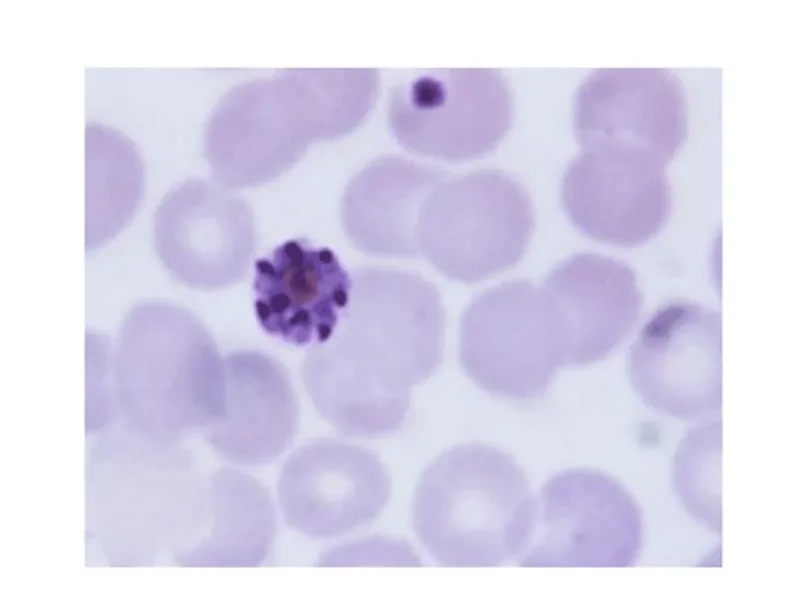
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and what results mean for patients.
April 20, 2026
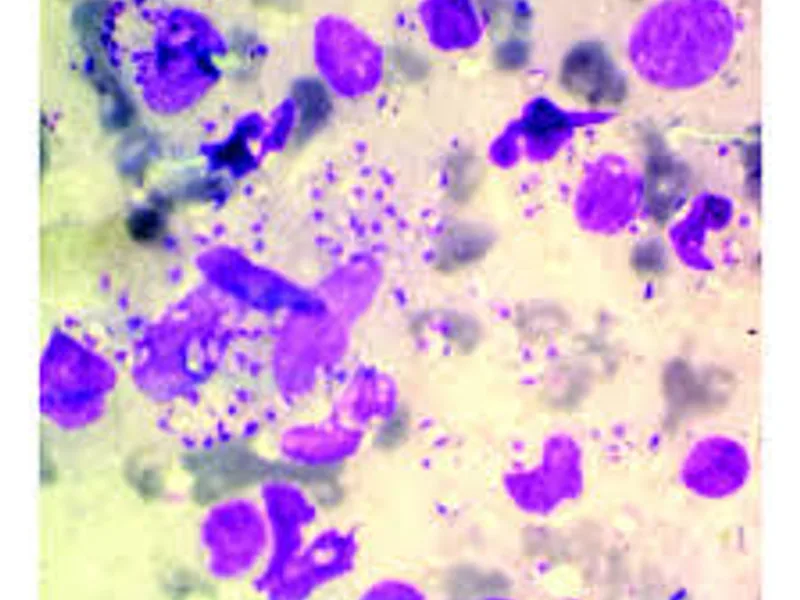
Discover how specialized staining techniques, like Giemsa, are crucial for accurately diagnosing Leishmaniasis, identifying the parasite, and guiding timely treatment. Understand this key diagnostic approach.
April 20, 2026
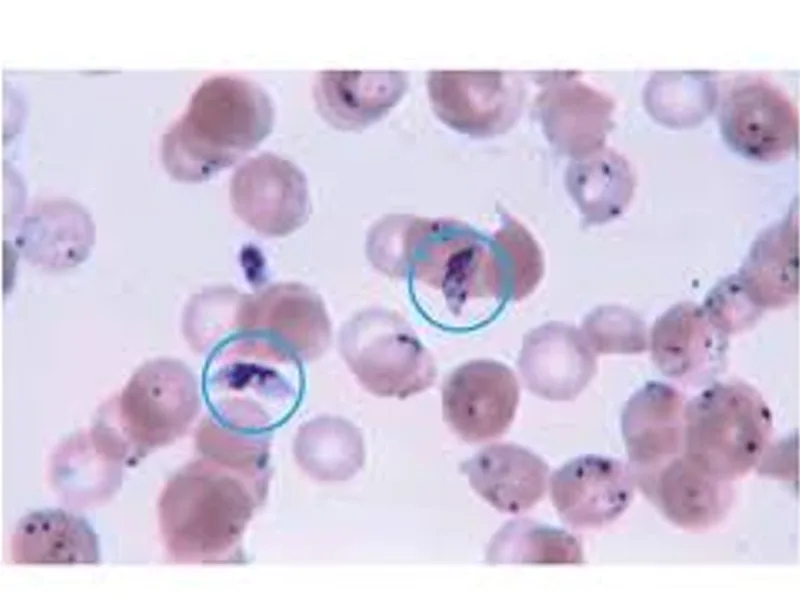
Understand the peripheral blood smear test for malaria diagnosis, its procedure, and why it's a vital tool in combating this disease.
April 20, 2026